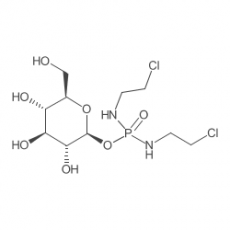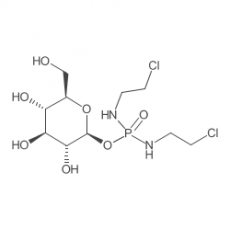
Glufosfamide
Glufosfamide, also known as glucophosphamide, D-glucose isophosphoramide mustard, D-19575, is a new, experimental cytotoxic chemotherapeutic agent for treatment of malignancies. Glufosfamide is, basically, a glycosidic conjugate between β-D-glucose and the active alkylating moiety of the well-known antineoplasic drug ifosfamide, so-called "isophosphoramide mustard". Glufosfamide combines the active part of ifosfamide, a member of a widely used class of chemotherapy drugs known as "alkylators," with a glucose molecule. Because of its glucose component and a tumor cell's increased need for glucose, glufosfamide may be preferentially transported into tumors compared to most normal tissues. Inside cells, the linkage between glucose and the alkylator is cleaved to release the active drug.
2: Mazur L, Opydo-Chanek M, Stojak M. Glufosfamide as a new oxazaphosphorine anticancer agent. Anticancer Drugs. 2011 Jul;22(6):488-93. doi: 10.1097/CAD.0b013e328345e1e0. Review. PubMed PMID: 21427562.
3: Chiorean EG, Dragovich T, Hamm J, Barrios CH, Gorini CF, Langmuir VK, Kroll S, Jung DT, Tidmarsh GT, Loehrer PJ. A phase 2 trial of glufosfamide in combination with gemcitabine in chemotherapy-naive pancreatic adenocarcinoma. Am J Clin Oncol. 2010 Apr;33(2):111-6. doi: 10.1097/COC.0b013e3181979204. PubMed PMID: 19687729.
4: Arafa HM. Possible contribution of beta-glucosidase and caspases in the cytotoxicity of glufosfamide in colon cancer cells. Eur J Pharmacol. 2009 Aug 15;616(1-3):58-63. doi: 10.1016/j.ejphar.2009.06.024. Epub 2009 Jun 21. PubMed PMID: 19545561.
5: Shimizu T, Okamoto I, Tamura K, Satoh T, Miyazaki M, Akashi Y, Ozaki T, Fukuoka M, Nakagawa K. Phase I clinical and pharmacokinetic study of the glucose-conjugated cytotoxic agent D-19575 (glufosfamide) in patients with solid tumors. Cancer Chemother Pharmacol. 2010 Jan;65(2):243-50. doi: 10.1007/s00280-009-1028-3. PubMed PMID: 19479254.
6: Ciuleanu TE, Pavlovsky AV, Bodoky G, Garin AM, Langmuir VK, Kroll S, Tidmarsh GT. A randomised Phase III trial of glufosfamide compared with best supportive care in metastatic pancreatic adenocarcinoma previously treated with gemcitabine. Eur J Cancer. 2009 Jun;45(9):1589-96. doi: 10.1016/j.ejca.2008.12.022. Epub 2009 Jan 31. PubMed PMID: 19188061.
7: Ammons WS, Wang JW, Yang Z, Tidmarsh GF, Hoffman RM. A novel alkylating agent, glufosfamide, enhances the activity of gemcitabine in vitro and in vivo. Neoplasia. 2007 Aug;9(8):625-33. PubMed PMID: 17786181; PubMed Central PMCID: PMC1950432.
8: Chiorean EG, Dragovich T, Hamm J, Langmuir VK, Kroll S, Jung DT, Colowick AB, Tidmarsh GF, Loehrer PJ. A Phase 1 dose-escalation trial of glufosfamide in combination with gemcitabine in solid tumors including pancreatic adenocarcinoma. Cancer Chemother Pharmacol. 2008 May;61(6):1019-26. Epub 2007 Jul 28. PubMed PMID: 17661038.
9: Sun YM, Chen XY, Zhong DF. [Identification of glufosfamide metabolites in rats]. Yao Xue Xue Bao. 2006 Jun;41(6):513-7. Chinese. PubMed PMID: 16927824.
10: Holzgrabe U. [Perfect drug targeting. Glufosfamide a new derivative]. Pharm Unserer Zeit. 2006;35(2):158-9. German. PubMed PMID: 16583570.
11: Sun Y, Chen X, Xu H, Guan Z, Zhong D. Stability of glufosfamide in phosphate buffers and in biological samples. J Chromatogr B Analyt Technol Biomed Life Sci. 2006 Mar 7;832(2):224-30. Epub 2006 Feb 7. PubMed PMID: 16455312.
12: Chen X, Sun Y, Cao X, Jin F, Zhong D. Determination of glufosfamide in rat plasma by liquid chromatography/tandem mass spectrometry. Rapid Commun Mass Spectrom. 2005;19(13):1839-43. PubMed PMID: 15945026.
13: Glufosfamide: beta-D-Glc-IPM, D 19575. Drugs R D. 2005;6(1):49-52. Review. PubMed PMID: 15801867.
14: Dollner R, Dietz A, Kopun M, Helbig M, Wallner F, Granzow C. Ex vivo responsiveness of head and neck squamous cell carcinoma to glufosfamide, a novel alkylating agent. Anticancer Res. 2004 Sep-Oct;24(5A):2947-51. PubMed PMID: 15517901.
15: Giaccone G, Smit EF, de Jonge M, Dansin E, Briasoulis E, Ardizzoni A, Douillard JY, Spaeth D, Lacombe D, Baron B, Bachmann P, Fumoleau P; EORTC-New Drug Development Group. Glufosfamide administered by 1-hour infusion as a second-line treatment for advanced non-small cell lung cancer; a phase II trial of the EORTC-New Drug Development Group. Eur J Cancer. 2004 Mar;40(5):667-72. PubMed PMID: 15010066.
16: van den Bent MJ, Grisold W, Frappaz D, Stupp R, Desir JP, Lesimple T, Dittrich C, de Jonge MJ, Brandes A, Frenay M, Carpentier AF, Chollet P, Oliveira J, Baron B, Lacombe D, Schuessler M, Fumoleau P; European Organization for Research and Treatment of Cancer New Drug Development Group; European Organization for Research and Treatment of Cancer Brain Tumor Group. European Organization for Research and Treatment of Cancer (EORTC) open label phase II study on glufosfamide administered as a 60-minute infusion every 3 weeks in recurrent glioblastoma multiforme. Ann Oncol. 2003 Dec;14(12):1732-4. PubMed PMID: 14630677.
17: Briasoulis E, Pavlidis N, Terret C, Bauer J, Fiedler W, Schöffski P, Raoul JL, Hess D, Selvais R, Lacombe D, Bachmann P, Fumoleau P. Glufosfamide administered using a 1-hour infusion given as first-line treatment for advanced pancreatic cancer. A phase II trial of the EORTC-new drug development group. Eur J Cancer. 2003 Nov;39(16):2334-40. PubMed PMID: 14556925.
18: Niculescu-Duvaz I. Glufosfamide (Baxter Oncology). Curr Opin Investig Drugs. 2002 Oct;3(10):1527-32. Review. PubMed PMID: 12431031.
19: Styczynski J, Wysocki M, Kurylak A, Juraszewska E, Malinowska I, Stanczak E, Płoszynska A, Stefaniak J, Mazur B, Szczepanski T, Ras M. In vitro activity of glufosfamide in childhood acute leukemia. Anticancer Res. 2002 Jan-Feb;22(1A):247-50. PubMed PMID: 12017297.
20: Briasoulis E, Judson I, Pavlidis N, Beale P, Wanders J, Groot Y, Veerman G, Schuessler M, Niebch G, Siamopoulos K, Tzamakou E, Rammou D, Wolf L, Walker R, Hanauske A. Phase I trial of 6-hour infusion of glufosfamide, a new alkylating agent with potentially enhanced selectivity for tumors that overexpress transmembrane glucose transporters: a study of the European Organization for Research and Treatment of Cancer Early Clinical Studies Group. J Clin Oncol. 2000 Oct 15;18(20):3535-44. PubMed PMID: 11032596.
| Chemical | |
|---|---|
| CAS Num | 132682-98-5 |
| Chemical Formula | C10H21Cl2N2O7P |
| Molecular Weight | 383.16 |
| IUPAC Chemical Name | N,N'-Bis(2-chloroethyl)phosphorodiamidic acid beta-D-glucopyranosyl ester |
| Exact Mass | 382.04634 |
| Elemental Analysis | C, 31.35; H, 5.52; Cl, 18.51; N, 7.31; O, 29.23; P, 8.08 |
| Synonym | D19575; D-19575; D 19575; glucophosphamide D-glucose; isophosphoramide mustard; Glufosfamide; glcIPM. |
| Solubility | Soluble in DMSO and soluble in water |
| SMILES Code | O=P(NCCCl)(NCCCl)O[C@H]1[C@@H]([C@H]([C@@H]([C@@H](CO)O1)O)O)O |
| Biological | |
| Targets and Effects | N/A |
| Pathways | alkylating agent |
| Physical | |
| Appearance | Solid powder |
| Purity | >98% (or refer to the Certificate of Analysis) |
| Shipping Condition | Shipped under ambient temperature as non-hazardous chemical. This product is stable enough for a few weeks during ordinary shipping and time spent in Customs. |
| Storage Condition | Dry, dark and at 0 - 4 C for short term (days to weeks) or -20 C for long term (months to years). |
| Shelf Life | >2 years if stored properly |
Mailing Address
Anba Pharma Company
3-H Gill Street, Suite 300
Woburn, MA 01801
United State
Our Contact Info.
Tel: 1+610-883-0668
Email: sales@AnbaPharma.com